Received: January 01, 1970
Published: January 01, 1970
Genet.Mol.Res. ():
Keywords
Preeclampsia, Adrenomedullin, Vasodilation, Gene polymorphisms.
Introduction
Preeclampsia (PE) is one of the frequently encountered obstetric complications of pregnancy which is associated with hypertension in conjunction with proteinuria and entails severe consequences such as seizures, kidney failure, haemorrhage in the mother (Sharon E et al., 2008; Sibai BM et al., 2003; ACOG 2002; Lindheimer MD et al., 2008) and intrauterine growth retardation (IUGR), intrauterine death (IUD) in fetus (Ounsted M et al., 1981, Mac Dorman, M. L et al., 2007). The global incidence of preeclampsia ranges between 5-14% and considered to be a leading cause of maternal and fetal/neonatal morbidity and mortality, if left untreated (Roberts, J.M. and Cooper, DW 2001). PE is characterised by inappropriate transformation of spiral arteries and impaired cytotrophoblasts differentiation that lead to shallow placentation in the early period of pregnancy. Further, defective remodelling of spiral arteries results in reduced utero placental perfusion, hypoxia thereby restrict fetal angiogenesis (Reynolds LP, Redmer DA 2001).
The etiology and patho-mechanism of PE remains poorly understood; however, the condition is believed to be associated with excessive maternal inflammation and endothelial dysfunction. Further, epidemiological and family studies have demonstrated a strong genetic contribution towards the susceptibility of this multifactorial disorder. Several polymorphic loci have been mapped by linkage analysis, nonetheless, only a few promising candidate genes have been identified so far. Adrenomedullin (ADM), a vasodilator peptide suggested to be one of the promising candidate genes identified to play a role in the establishment and maintenance of healthy pregnancy. The gene is expressed in most organs including reproductive tissues such as uterine endometrium, fetal membranes, placenta, endometrial stromal macrophages, trophoblasts cells, endothelial cells and vascular smooth muscle cells. (Hague S, et al., 2000, Trollmann R, et al., 2002, Minegishi T, et al., 1999, Zhao Y, et al., 1998, Gratton RJ, et al., 2003, Karpinich NO et al., 2011, Li M et al., 2008).
Plasma concentrations of ADM are elevated two to three fold above baseline levels in various diseases and intriguingly largest in normal pregnancies. However, low maternal serum levels of ADM are reported to be allied with PE (Boc-Zalewska A, et al., 2011). ADM gene expression and its polymorphic variants are being investigated as probable risk factors of preeclampsia and gestational hypertension development in various studies (Li M et al., 2008; Lahiri D K and Nurnberger J I, Jr et al., 1991; Ishimitsu T et al., 2006). Keeping the above information in view, it can be cogitated that genetic variants that alter the ADM levels may be important in pregnancy related pathogenesis such as PE. Hence, the present study is aimed to investigate two polymorphic variants (rs4399321A/G and rs4910118C/T) that reside on ADM gene among PE patients and healthy controls in South Indian subjects.
Materials and Methods
Sampling
The present study was conducted on a total of 600 samples comprising of 300 subjects with Pre-eclampsia and 300 healthy pregnant women (control subjects) visiting Government Maternity Hospital, Hyderabad and Gandhi Hospital, Hyderabad, Telangana, India. This study was approved by the Institutional Ethical Review Board, Osmania University, Hyderabad, Telangana, India, and all the subjects provided written informed consent for their inclusion in the investigation. The study was conducted during the period 2010-2013, with the information collected from all the participants on demographic, clinical, and family history of PE in a well-designed proforma for the analysis.
Inclusion criteria
The diagnosis of PE was conducted in accordance with the International Society for the Study of Hypertension in Pregnancy [ISSHP] which defines PE as blood pressure readings of at least 140/90 mmHg on two occasions 6 hrs apart after 20 weeks of gestation, together with proteinuria ≥ 300 mg in a 24 hrs urine collection or, ≥ 1+ on dipstick testing not associated with urinary tract infection or ruptured membranes. Severe PE was defined as blood pressure ≥ 160/110 mmHg or proteinuria ≥ 2 g/24 hrs or at least 3+ reading on dipstick in a random urine specimen after 20 weeks of pregnancy. Mild PE was defined as blood pressure ≥ 140/90 mmHg and proteinuria ≥ 300 mg/24 hrs or ≥ 1+ reading on dipstick in a random urine specimen after 20 weeks of pregnancy. Ethnically matched normotensive pregnant women in the third trimester of pregnancy, with blood pressure ≤ 120/80 mmHg, no proteinuria and no history of hypertension were included as normal healthy control subjects.
Exclusion criteria
Individuals with chronic hypertension, diabetes, polycystic ovary syndrome, multiple gestations, premature preterm rupture of membranes or with unexplained vaginal bleeding were excluded from the study. Likewise, subjects who were on antihypertensive medication and individuals who conceived by assisted reproductive technology were also excluded from the study.
Molecular analyses
After collecting 3 ml of peripheral venous blood in EDTA coated vials, Genomic DNA was extracted by standard protocol (Lahiri DK and Nurnberger JI, Jr. et al., 1991), purity was checked, and stored at -20°C for further molecular analysis. PCR amplification of the selected polymorphisms (rs4399321A/G and rs4910118C/T) was carried out in a total volume of 10úl reaction mixture that includes 1.5 μl of 100 ng DNA, 1 μl PCR buffer, 1-1.5 μl of each primers, 2.5 μl dNTPs 1.5 μl of Taq DNA polymerase under thermal conditions of 95°C for 5 min; 35 cycles of 95°C for 30 seconds, annealing temperature at 51°C for rs4399321 A/G and 51.5°C for rs4910118C/T and initial extension at 72°C for 30 seconds with a final extension at 72°C for 10 min.
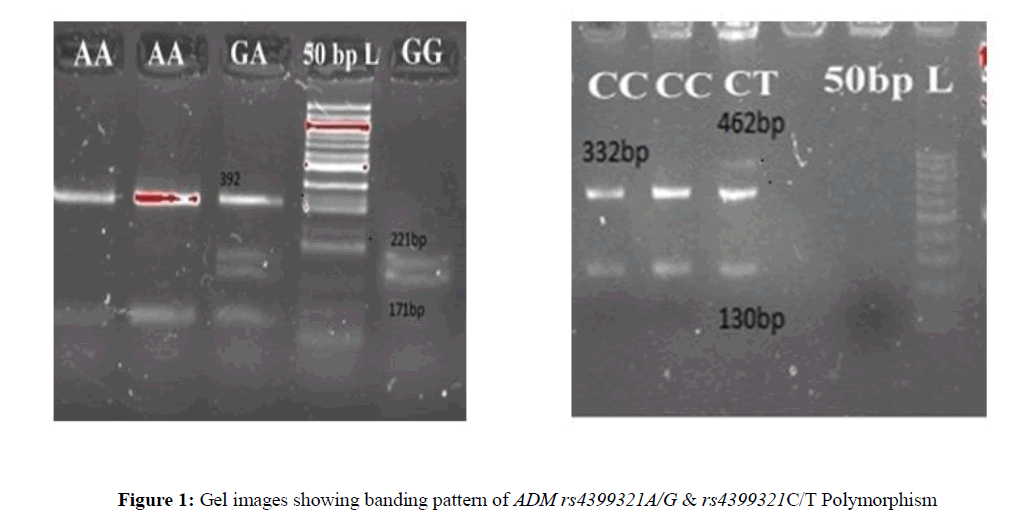
The PCR products of rs4399321A/G were digested with Hae III enzyme at a final concentration of 0.15U/ l for 2 hours at 37°C in dry bath and consequently electrophoresed on a 2.5% agarose gel at 100 V, visualized under UV light using gel doc (Bio-Rad). Genotypes were analyzed by fragment sizes; the AA genotype was documented by the presence of 221 bp bands, heterozygous AG genotype by the presence of 221 bp, 192 bp, 171 bp bands and homozygous GG genotype by the presence of 192 bp, 171 bp. Similarly, the PCR products of rs4910118C/T were digested with 0.2 U/l of BtgI restriction endonuclease for 2 hours at 37°C in dry bath and electrophoresed on a 2.5% agarose gel. The CC genotype was identified by the presence of 462 bp, heterozygote CT genotype by the presence of 462 bp, 332 bp, 130 bp and homozygous TT genotype by the presence of 332 bp, 130 bp (Figure 1).
Statistical Analysis
Descriptive statistics were expressed in mean ± standard deviation [SD]. Clinico-demographic characteristics were compared using the Student’s t-test or Z-test for proportions as applicable. A chi-square test was performed to calculate the differences in the allele and genotype frequency of the patient and control groups. The Hardy-Weinberg equilibrium (HWE) was tested using online calculator by Michael H Court to see the association between observed and expected genotype frequencies in cases and controls. The genotype risk towards the disease was determined by odds ratio (OR) calculator with its corresponding 95% confidence interval (CI). Haplotype frequency was performed by using online tool SNP stats and pairwise linkage disequilibrium (LD) analysis was done using Haploview software (version 4.2). Data analysis was carried out by SPSS version 21. A two-sided p-value of <0.05 was considered statistically significant.
Results
The demographic and clinical characteristics of the study population are shown in Table 1. The mean age at onset of pre-eclamptic patients was recorded as 22.3 ± 3.66 years. The mean proteinuria of the patients that was observed with dipstick was reported as 2.9 ± 0.94 mg/dl. The mean values of maternal age, gestational age, age at marriage, years of marriage, area of living, occupation, socio-economic status, education and blood pressure varied significantly between the patient and control groups (p<0.05). Further the mean systolic and diastolic blood pressures also differed significantly between the cases and controls. No significant difference was seen with respect to Gravidity among controls and patients (p>0.05). We observed 21% of IUGR & IUD cases, 10% of recurrent cases, and 4.6% of familial cases in preeclamptic group. Significant variation was observed in relation to family history of hypertension (p<0.05) whereas no difference was noted with family history of diabetes between the two groups (p>0.05).
| Variables | Controls (300) N (%) Mean ± SD |
Patients (300) N (%), Mean ± SD |
p-value |
|---|---|---|---|
| Age at onset (years) Proteinuria (mg/dl) Age at sample collection (years) Gestational age (weeks) Age at marriage (years) ≤18years >18years Years of marriage (years) ≤10years >10years Area of living Rural Urban Occupation Yes No Socioeconomic status Low High Education Illetrate Literate Systolic diastolic pressure (mmHg) Diastolic blood pressure (mmHg) Gravida 1 >1 Pregnancy complications (IUGR& IUD), Recurrence cases Non-recurrence cases Familial cases Non familial cases Family history of hypertension No family history of hypertension Family history of Diabetics Family history of Diabetics |
- <0.3 22.3 ± 3.0 35.7 ± 1.73 19.5 ± 2.70 109 (36) 17.04 ± 1.15 191 (63) 20.8 ± 2.31 3.1 ± 2.5 295 (98.3) 5 (1.6) 163 (54) 137 (45) 77 (25) 223 (74) 178 (59) 122 (40) 140 (46) 160 (53) 156.6 ± 13.7 79 ± 3.60 166 (55) 139 (46) - - - - - 15 (5%) 285 (95%) 20 (6.6%) 280 (93.3%) |
22.1 ± 3.7 2.9 ± 0.94 22.9 ± 3.8 29.8 ± 4.73 18.8 ± 2.95 139 (46)16.6 ± 1.48 162 (54)21.0 ± 2.24 4.03 ± 3.9 271 (90) 29 (9.6) 190 (63) 110 (36) 162 (54) 138 (46) 225 (75) 75 (25) 168 (56) 132 (44) 118.42 ± 3.90 102.4 ± 9.5 181 (60) 119 (39) 63 (21%) 30 (10%) 270 (90%) 14 (4.6%) 286 (95.5%) 51 (17%) 259 (86.6%) 11 (3.6%) 289 (86.6%) |
<0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 <0.05 >0.05 >0.05 - - - - - <0.05 <0.05 >0.05 >0.05 |
*p<0.05; #p<0.10
Table 1: Clinical and demographic characteristics of preeclamptic patients and normal pregnant women.
Genotyping of ADM rs4399321A/G polymorphism
The genotype and allele frequencies for the control and the patient groups are presented in Table 2. The genotype distribution of AA, AG, GG in patients were 28%, 70%, 1.3% and healthy pregnant women were 40%, 63.3% and 0% respectively. The genotypes varied significantly between the patients and controls (χ2=10.8, p<0.05), whereas the allele frequencies did not differ between the two groups (χ2=1.44; p>0.05).
| rs4399321A/G | AA N (%) |
AG N (%) |
GG N (%) |
A | G | Group Comparison | OR (95% CI) | p-value | |
|---|---|---|---|---|---|---|---|---|---|
| Controls (300) Patients (300) |
105 (35) 82 (27.3) |
195 (65) 214 (71.3) |
- 4 (1.3) |
0.63 0.71 |
0.37 0.29 |
AA vs. others AG vs. others GG vs. others A vs. G G vs. A |
0.69 (0.49-0.98) 1.33 (0.94-1.89) - 0.69 (0.38-1.25) 1.43 (0.79-2.60) |
0.05 0.11 0.29 0.29 |
|
| χ2= 10.8; p (0.02) | χ2=1.14; p (>005) | ||||||||
| HWE | Controls | χ2=69.5; p=0.0000 | |||||||
| Patients | χ2=84.3 ; p=0.0000 | ||||||||
| rs4910118C/T | CC N (%) |
CT N (%) |
TT N (%) |
C | T | Group Comparison | OR (95% CI) | p-value | |
| Controls (300) Patients (300) |
180 (60) 205 (68) |
120 (40) 95 (32) |
0 0 |
0.84 0.80 |
0.16 0.20 |
CC vs. others CT vs. others C vs. T T vs. C |
1.43 (1.02-2.01) 0.69 (0.49-0.97) 1.31 (0.63-2.71) 0.76 (0.36-1.57) |
0.04 0.04 0.58 0.58 |
|
| χ2= 4.53 ; p=0.0 | χ2=0.54 ; p >0.05 | ||||||||
| HWE | Controls | χ2=18.7 ; p=0.000015 | |||||||
| Patients | χ2= 15.61 ; p=0.00007 | ||||||||
| *p<0.05; #p<0.10 | |||||||||
*p<0.05; #p<0.10
Table 2: Genotype and allelic frequency distribution of ADM rs4399321 A/G & rs4910118 C/T polymorphisms among PE patients and Healthy pregnant women
The genotype frequencies deviated from HW equilibrium in both cases and controls (controls χ2=69.5; patients χ2=84.3; p<0.05). Individuals carrying the AA genotype were higher in controls (40%) compared to patients (28%) and revealed an OR value of 0.69 (CI 0.49-0.98; p=0.05). MLR analysis showed lack of association of rs4399321 A/G polymorphism when adjusted with age, years of marriage, gravida, gestational age, blood pressure, recurrence and complications, however, appreciable association was observed with the familial cases (p=0.06).
Genotyping of rs4910118 C/T polymorphism
The genotype and allele distribution for the healthy normal pregnant women and the preeclamptic patients group of this polymorphism are presented in Table 2. The frequency distribution of CC and CT genotypes was noted to be 68% and32% in preeclampsia patients whereas controls revealed a frequency of 60% and40% respectively; we did not find TT genotype in both the groups. The distribution of genotypes differed significantly between the patients and controls (χ2=4.53; p<0.05), whereas the allele frequencies did not (χ2=0.54; p>0.05). Individuals carrying CC genotype predominated in the patients with an OR of 1.43 (CI 1.02–2.01, p=0.04), whereas, CT vs others gave an OR value of 0.69 (CI 0.49–0.97; p=0.04). The genotype frequencies are not in consistent with HW equilibrium in both control and patient groups (p<0.05).
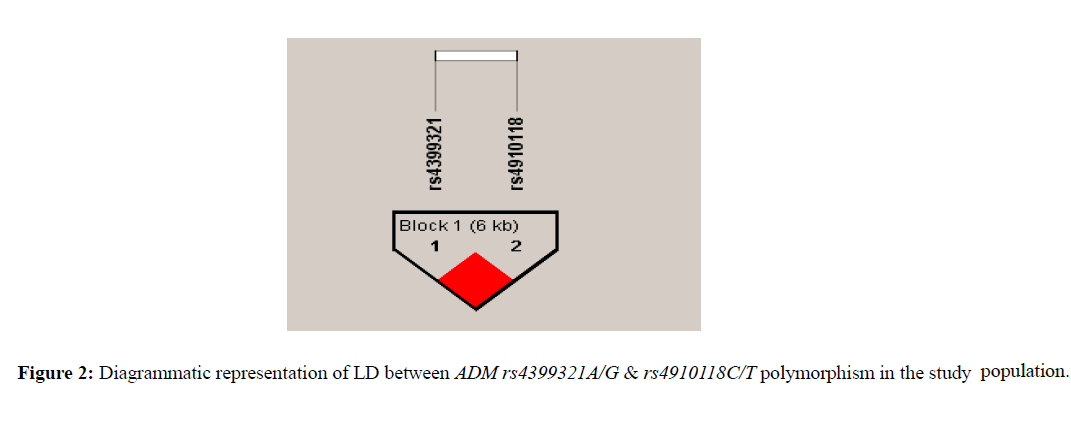
MLR analysis revealed no association of rs4910118 C/T polymorphism after adjusted with various parameters such as age, years of marriage, gravida, gestational age, blood pressure, family history, recurrence and complications. Further, haplotype analysis exhibited the A-T haplotype block frequency to be significantly elevated in controls than patients with an OR of 0.40 (CI 0.21-0.75, p=0.004). A strong Linkage disequilibrium LD was observed between the two polymorphic markers of the ADM gene (D’=1.0 and r2=0.12, p<0.05) (Figure 2 and Table 3).
| S. No | 3120 A/G |
175 C/T | Group 0 | Group 1 | Over all frequency | OR (95% CI) | p-value |
|---|---|---|---|---|---|---|---|
| 1 | A | C | 0.5 | 0.6 | 0.5497 | 1.00 | ---- |
| 2 | G | C | 0.3 | 0.3 | 0.8208 | 1.43 (0.95-2.16) | 0.09 |
| 3 | G | T | 0.2 | 0.01 | 0.9361 | 2.64 (0.75-9.33) | 0.13 |
| 4 | A | T | 0 | 0.1 | 1 | 0.40 (0.21-0.75) | 0.0043 |
Table 3: Haplotype analysis of ADM rs4399321 A/G & rs4910118 C/Tpolymorphisms between healthy pregnant women and preeclampsia patients.
Discussion
Adrenomedullin is a novel vasoactive peptide hormone was first isolated from human pheochromocytoma (Kitamura K et al., 1993). It belongs to the amylin/calcitonin gene-related peptide (CGRP) super-family located on chromosome 11 (11pl5.1-3) and comprised of four exons and three introns (Beltowski J, Jamroz A 2004; Takei Y et al., 2004; Roh J et al., 2004, Ishimitsu T et al., 2001a). ADM expression is regulated by several factors involved in the physiology of reproduction. It was speculated that ADM is an important molecule in preventing an immune attack at peri-implantation period (Lenhart PM et al., 2014). Trophoblastic giant cells express high levels of ADM throughout pregnancy which was demonstrated in animal studies and these cells are active players in the process of implantation, decidualization and placentation (Hata T, et al., 1997). ADM+/- female mice revealed that the expression and dosage of maternal ADM is a critical determinant for establishing uterine receptivity (Li M et al., 2006).
Genetic variations in the ADM gene have been related with some diseases including preeclampsia (Gratton RJ, et al., 2003) and may possibly be accountable for the differences in serum quantitative estimates with respect to adrenomedullin. Few of these polymorphisms have shown to be linked with altered ADM mRNA expression and increased incidence of gestational hypertension and preeclampsia (Ishimitsu T et al., 2006) providing further evidence for the probable role of ADM in the pathogenesis of pregnancy related conditions. In addition, signalling between the adrenomedullin and its receptor has a considerable role in placental development (Witlin AG et al., 2002).
Among the nucleotide variants identified in the ADM gene, the two Single Nucleotide Polymorphisms (SNPs) gained attention are, the rs4399321 A/G located in the 5’UTR region of ADM gene, which enhances adrenomedullin transcription (Shufeng Chen et al., 2013), and rs4910118 C/T located at 762 bp downstream of the 3’ end of ADM gene which is involved with reduced adrenomedullin transcription (Yujin Kobayashi et al., 2005). However, no previous reports that assessed the relationship between polymorphic variants of ADM and pre-eclampsia in south Indian women exist. Hence, we attempted to explore the relationship between rs4399321A/G and rs4910118C/T and PE susceptibility in our population.
To the best of our knowledge, our study is the first to evaluate the selected 5’UTR (rs4399321A/G) and 3’UTR (rs4910118C/T) adrenomedullin gene polymorphisms in relation to the reproductive disorder, preeclampsia. The wild type A allele of rs4399321A/G was observed to be in elevated frequency by 5% among normotensive pregnant women compared to the preeclamptic patients. There are no relevant studies pertaining to this polymorphism in the literature to compare our results. However, a study from Chinese population conducted by Chen et al., (2013) showed an augmented frequency of A allele in individuals with lower systolic blood pressure compared to hypertensive groups suggesting the regulatory role for ADM in blood pressure homeostasis in humans (Shufeng Chen et al., 2013). Another study found no association of this polymorphism with essential hypertension (Yujin Kobayashi et al., 2005). Further, studies on knockout mice revealed higher renal perfusion pressure in ADM+/- mice than the wild type mice and lowest renal perfusion pressure in mice with over expressing adrinomodullin. Based on the circulating concentrations of ADM, it was reported that a largest increase in ADM levels was observed during healthy pregnancy, (Gratton RJ, et al. 2003). This prompt us to speculate that the A allele which is over represented in our normal pregnant group might be the one which is associated with higher expression of ADM compared to the counterpart allele. Moreover, our Insilico analysis revealed that rs4399321 is adjacent to a tract of polypyrimidine motif containing CPYnG consensus sequence whose alteration may result into upregulated translation. Estimation of adrenomedullin levels in relation to this polymorphism through cell based studies may establish the functional significance of this polymorphism.
With respect to the rs4910118 C/T polymorphism, substantial difference was seen in the genotype frequencies among the pre-eclampsia patients and normotensive pregnant women. Significantly increased frequency of homozygous CC genotype in PE patients than in controls suggests the predisposing role of this genotype towards PE (OR=1.43; 1.02-2.01; p=0.04). On the other hand, enhanced frequency of heterozygous CT genotype among normotensive pregnant women (OR=0.69; 0.49-0.97: p=0.04) is indicative of heterozygous advantage for the normal pregnancy and in other way provides safeguard against PE. The carriers of minor allele (T) of this single nucleotide polymorphism was observed to have a mean plasma ADM levels that was 10.5% lower than in the non-carriers of this allele and shown to be independently associated with plasma ADM levels in cardiovascular cohort group from Hong-kong (Cheung BM,et al., 2011). In the absence of clear information on the concentrations of ADM in relation to this polymorphism and based on the above information, one can cautiously infer that heterozygotes of this polymorphism may be associated with intermediate levels of adriomeddulin that provide congenial environment for the maintenance of normal pregnancy. Auxiliarily, our insilico analysis carried out by using RegRNA tool revealed that rs4910118C/T region corresponds to the Exon splicing enhancer-SRp40 protein binding site which needs to be clarified further (RegRNA 1.0 version).
It is interesting to note that the results of individual SNP were also reflected in haplotype analysis. The haplotype block A-T showed a significant protection compared to other haplotype blocks suggesting the protective role of A/G polymorphism of ADM gene against preeclampsia, which further strengthened by strong LD between these two SNPs ( (D’=1.0 and r2=0.12, p<0.05).
Logistic regression analysis revealed none of the variables such as age, years of marriage, gravida, gestational age, blood pressure, familial, recurrence and pregnancy related complications such as IUGR & IUD, to be associated with the selected polymorphisms (Tables 4 and 5).
| Variables | AA vs. others | p-value | GA vs. others | p-value | GG vs. others | P -value |
|---|---|---|---|---|---|---|
| Age | 0.54 (0.25-1.18) | 0.12 | 1.63 (0.76-3.47) | 0.20 | 5.41 (0.42-68.1) | 0.19 |
| Years of marriage | 1.37 (0.49-3.81) | 0.54 | 0.80 (0.29-2.22) | 0.68 | 0.00 (0.000 | 0.99 |
| Gravida | 0.69 (0.38-1.23) | 0.21 | 1.27 (0.71-2.27) | 0.40 | 68264.71 (0.0000) | 0.99 |
| Gestational age | 1.23 (0.70-2.15) | 0.46 | 0.86 (0.50-1.49) | 0.61 | 0.36 (0.04-3.04) | 0.35 |
| Blood pressure | 1.08 (0.57-2.07) | 0.79 | 0.82 (0.43-1.56) | 0.56 | 87153.6741 (0.000) | 0.99 |
| Familial | 0.72 (0.19-2.72) | 0.63 | 0.98 (0.28-3.23) | 0.95 | 12.9 (0.87-190.16) | 0.06 |
| Recurrence | 0.47 (0.16-1.39) | 0.17 | 2.13 (0.73-6.18) | 0.16 | 0.0000 (0.000) | 0.0993 |
| Complications | 1.09 (0.58-2.06) | 0.76 | 0.98 (0.52-1.84) | 0.97 | 0.000 (0.000) | 0.9990 |
Table 4: Multiple logistic regression analysis (ADM rs4399321 A/G polymorphism vs. Risk factors)
| Variables | CC vs CT + TT | p-value | CT vs TT+ CC | p-value |
|---|---|---|---|---|
| Age | 0.77 (0.39-1.52) | 0.45 | 1.63 (0.76-3.47) | 0.20 |
| Years of marriage | 0.54 (0.21-1.35) | 0.19 | 0.80 (0.29-2.22) | 0.80 |
| Gravida | 1.08 (0.61-1.92) | 0.77 | 1.27 (0.71-2.27) | 0.40 |
| Gestational age | 1.09 (0.64-1.84) | 0.74 | 0.86 (0.50-1.49) | 0.61 |
| Blood pressure | 0.65 (0.35-1.24) | 0.65 | 0.82 (0.43-1.56) | 0.56 |
| Familial | 0.74 (0.23-2.33) | 0.60 | 0.96 (0.28-3.23) | 0.95 |
| Recurrence | 1.22 (0.49-3.04) | 0.66 | 2.13 (0.73-6.18) | 0.16 |
| Complications | 1.25 (0.66-2.37) | 0.48 | 0.98 (0.52-1.84) | 0.97 |
Table 5: Multiple logistic regression analysis (ADM rs4910118 C/T polymorphism vs. risk factors)
The understanding of the role of ADM through human studies is limited and inconsistent. The present study was an attempt to see the role of ADM genetic polymorphisms with PE as there is lack of information from this part of the subcontinent. Our study with fairly good sample size revealed an association of selected gene polymorphisms with PE, however, the limitation being scarce of supportive literature on circulating levels of ADM to compare with respective genotypes. Future human studies dealing with the genotype- phenotype correlation and downstream effect of ADM on the placental hypoxic tissue may help in developing medically relevant molecular markers in normal pregnancy and its complications.
Authors' Contributions
Goske Deepthi performed the experimentation and drafted the manuscript. Komaravalli Prasanna Latha applied statistics and gave valuable suggestions for drafting the manuscript. Vital Usharani provided samples for the study. Police Fazul Rahman read the manuscript and offered valuable inputs. Parveen Jahan conceived and envisioned the experiment, prudently read manuscript and included critical suggestions.
Acknowledgments
We extend our thanks to University Grants Commission [UGC], New Delhi, India for financial support [F.No.34-217/2008 (SR)]. We are thankful to all the patients and controls enrolled in the present study for their cooperation in providing blood samples and clinical information.
Conflict of Interest
The authors have declared that no competing interests exist.
About the Authors
References
- American College of Obstetricians and Gynecologists (2002) Practice bulletin: Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol 99: 159-167. https://doi.org/10.1097/00006250-200201000-00028
- Beltowski J and Jamroz A (2004) Adrenomedullin–what do we know 10 years since its discovery? Pol J Pharmacol 56: 5-27.
- Bernard MY, Cheung, Kwok Leung Ong, Annette WK Tso, Raymond YH Leung, et al. (2011) Plasma adrenomedullin level is related to a single nucleotide polymorphism in the adrenomedullin gene. European journal of endocrinology 165: 571-577. https://doi.org/10.1530/eje-11-0513
- Boc-Zalewska A (2011) Adrenomedullin mRNA expression in placenta of preeclamptic women. Ginekol Pol. 82: 585-591.
- Gratton RJ, et al. (2003) Adrenomedullin messenger ribonucleic acid expression in the placentae of normal and preeclamptic pregnancies. J Clin Endocrinol Metab 88: 6048-6055. https://doi.org/10.1210/jc.2003-030323
- Hague S, et al. (2000) Expression of the hypoxically regulated angiogenic factor adrenomedullin correlates with uterine leiomyoma vascular density. Clin Cancer Res. 6: 2808-2814.
- Hata T, et al. (1997) Decreased circulating adrenomedullin in pre-eclampsia. Lancet 350: 1600. https://doi.org/10.1016/s0140-6736 (05)64016-0
- Ishimitsu T, Hosoya K, Tsukada K, Minami J, et al. (2001) Chromosomal sublocalization and microsatellite DNA polymorphism of human adrenomedullin gene. Peptides 22:1739-1744. https://doi.org/10.1016/s0196-9781 (01)00531-9
- Ishimitsu T, Ono H, Minami J et al. (2006) Pathophysiologic and therapeutic implications of adrenomedullin in cardiovascular disorders, Pharmacol Ther 111: 909- 927. https://doi.org/10.1016/j.pharmthera.2006.02.004
- Karpinich NO, Hoopes SL, Kechele DO, Lenhart PM, et al. (2011) Adrenomedullin function in vascular endothelial cells: insights from genetic mouse models. Curr Hypertens Rev 7: 228-239. https://doi.org/10.2174/157340211799304761
- Kitamura K, Kawamoto M, Ichiki Y, Nakamura S, et al. (1993) Adrenomedullin: A novel hypotensive peptide isolated from human pheochromocytoma. Biochem Biophys Res Commun 192: 553-560. https://doi.org/10.1006/bbrc.1993.1451
- Lahiri D K and Nurnberger J I (1991) A rapid non-enzymatic method for the preparation of HMW DNA from blood for RFLP studies. Nucleic Acids Res 19: 5444. https://doi.org/10.1093/nar/19.19.5444
- Lenhart PM, Nguyen T, Wise A, Caron KM, et al. (2014) Adrenomedullin signaling pathway polymorphisms and adverse pregnancy outcomes. Am J Perinatol 31: 327-334. https://doi.org/10.1055/s-0033-1349345
- Li M, Wu Y, Caron KM (2008) Haploinsufficiency for adrenomedullin reduces pinopodes and diminishes uterine receptivity in mice. Biol Reprod 79: 1169-1175. https://doi.org/10.1095/biolreprod.108.069336
- Li M, Yee D, Magnuson TR, Smithies O, et al. (2006) Reduced maternal expression of adrenomedullin disrupts fertility, placentation, and fetal growth in mice. J Clin Invest 116: 2653-2662. https://doi.org/10.1172/jci28462
- Lindheimer MD, Taler SJ, Cunningham FG (2008) Hypertension in pregnancy. J Am Soc Hypertens 2: 484-494. https://doi.org/10.1016/j.jash.2008.10.001
- MacDorman, ML Munson, S Kirmeyer (2007) “Fetal and perinatal mortality, United States, 2004,” National Vital Statistics Reports. 56: 1-19.
- Minegishi T (1999) Adrenomedullin and atrial natriuretic peptide concentrations in normal pregnancy and pre-eclampsia. Mol Hum Reprod 5: 767-770. https://doi.org/10.1093/molehr/5.8.767
- Ounsted M, Moar V, Scott WA (1981) Perinatal morbidity and mortality in small-for-dates babies: the relative importance of some maternal factors. Early Hum Dev 5: 367-375. https://doi.org/10.1016/0378-3782 (81)90017-7
- Reynolds LP and RedmerDA (2001) Angiogenesis in the placenta. Biol Reprod 64:1033-1040. https://doi.org/10.1095/biolreprod64.4.1033
- Roberts JM and Cooper DW (2001) Pathogenesis and Genetics of Preeclampsia. Lancet 357: 53-56. https://doi.org/10.1016/s0140-6736 (00)03577-7
- Roh J, Chang CL, Bhalla A, Klein C, et al. (2004) Intermedin is a calcitonin/calcitonin gene-related peptide family peptide acting through the calcitonin receptor-like receptor/receptor activity-modifying protein receptor complexes. J Biol Chem 279: 7264-7274. https://doi.org/10.1074/jbc.m305332200
- Sharon E Maynard, S Ananth Karumanchi (2008) Molecular and Genetic Basis of Renal Disease. Clinical Features and Epidemiology 441-451.
- Shufeng Chen, Xiangfeng Lu, Qi Zhao, Laiyuan Wang, et al. (2013) Association of adrenomedullin gene polymorphisms and blood pressure in a Chinese population. Hypertension Research 36: 74-78. https://doi.org/10.1038/hr.2012.132
- Sibai BM, Caritis S, Hauth J (2003) What we have learned about preeclampsia Semin Perinatol 27: 239-246. https://doi.org/10.1016/s0146-0005 (03)00022-3
- Takei Y, Inoue K, Ogoshi M, Kawahara T, et al. (2004) Identification of novel adrenomedullin in mammals: a potent cardiovascular and renal regulator. FEBS Lett 556: 53-58. https://doi.org/10.1016/s0014-5793 (03)01368-1
- Trollmann R (2002) Adrenomedullin gene expression in human placental tIssue and leukocytes: a potential marker of severe tIssue hypoxia in neonates with birth asphyxia. Eur J Endocrinol 147: 711-716. https://doi.org/10.1530/eje.0.1470711
- Witlin AG, Li ZY, Wimalawansa SJ, Grady JJ, et al. (2002) Placental and fetal growth and development in late rat gestation is dependent on adrenomedullin. Biol Reprod 67:1025-1031. https://doi.org/10.1095/biolreprod.101.002196
- Yujin Kobayashi, Tomohiro Nakayama, Naoyuki Sato, Yoichi Izumi, et al. (2005) Haplotype-Based Case-Control Study Revealing an Association between the Adrenomedullin Gene and Proteinuria in Subjects with Essential Hypertension. Hypertension Research 28: 229-236. https://doi.org/10.1291/hypres.28.229
- Zhao Y, et al. (1998) PCR display identifies tamoxifen induction of the novel angiogenic factor adrenomedullin by a non-estrogenic mechanism in the human endometrium. Oncogene 16: 409-415. https://doi.org/10.1038/sj.onc.1201768
Download:
Full PDF- Share This