Abstract
Protein expression and characterization of SEP3 from Arabidopsis thaliana
Author(s): Q. Shi, J. Zhou, P. Wang, X. Lin and Y. XuSEPALLATA (SEP) MADS-box genes play crucial roles in the regulation of floral growth and development. They are required for the specification of sepals, petals, stamens, and carpels as well as for floral determinacy. SEPs perform their functions through the formation of homo- or hetero-polymers, which are the molecular basis of floral quartets. In vitro assays indicated that SEP3 forms a tetramer after binding to DNA, but it is unclear whether DNA binding induces the tetramer, because SEP3 is often reported to form a dimer. Here, we analyzed the oligomeric status of SEP3 domains in the absence of the DNA-binding MADS-box domain. The truncated SEP3 was constructed as a fusion protein and expressed in prokaryotic cells. The purified protein fragment displayed as a tetramer in the size exclusion chromatographic column, and a glutaraldehyde cross-linking assay demonstrated that the protein contained a dimer unit. Yeast two-hybrid tests further verified that the fragments form homologous polymers in vivo, and that the K domain is involved in tetramer formation. Current results imply that the SEP3 protein regulates the formation of flower meristems using the tetramer as a unit, and that the DNA-binding MADS-box is dispensable for polymer formation. The C-terminal region does not contribute to homo-tetramer formation, but it may be reserved to glue other proteins.
Impact Factor an Index

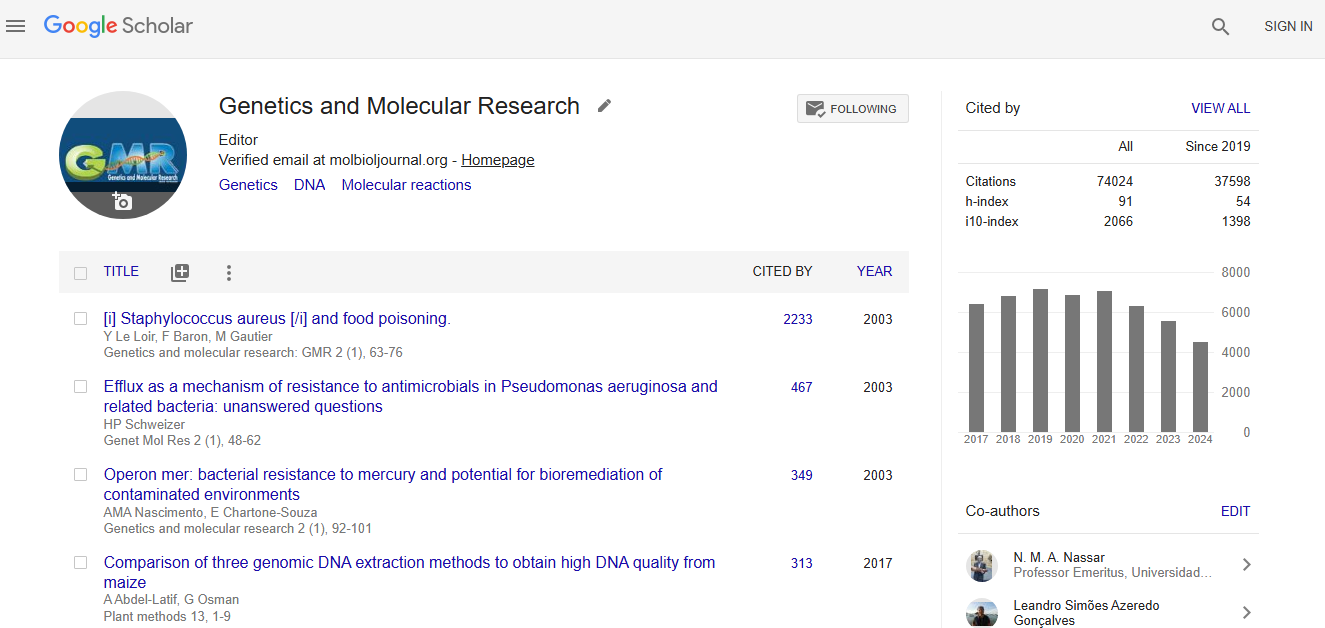
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report