Abstract
Protective effect of interleukin-10 and recombinant human keratinocyte growth factor-2 on ventilation-induced lung injury in rats
Author(s): J.Y. Wu, Z.H. Xiong, G.Z. Xiong, F.Q. Ding, J. Lei, S. Lu, Y. Li, G.M. He, L.L. Zhao and Z.J. LiuA rat model of ventilation-induced lung injury (VILI) during anesthesia was generated to investigate the potential role and possible mechanism of interleukin-10 (IL-10) and recombinant human keratinocyte growth factor-2 (rhKGF-2) in protecting anesthetized rats against VILI. A total of 50 male SD rats were randomly divided into 5 groups (N = 10 each): control, VILI, IL-10, rhKGF-2, and IL-10 + rhKGF-2. The VILI (model) group was generated via ventilation, with a tidal volume of 20 mL/kg. Rats in the IL-10 and rhKGF-2 groups received 8 mg/kg IL-10 and 5 mg/kg rhKGF-2, respectively, prior to ventilation. The rats in the IL-10 + rhKGF-2 group received both 8 mg/kg IL-10 and 5 mg/kg rhKGF-2 72 h before ventilation. The total number of nucleated cells and neutrophils in the bronchial alveolar lavage fluid was quantified, and the pathological changes in the pulmonary tissues examined by hematoxylin and eosin staining. The transcript and protein levels of surfactant protein C (SP-C) in lung tissues were detected by real-time polymerase chain reaction and western blot analyses. The SP-C mRNA expression in both IL-10 and rhKGF-2 groups was similar to that in the VILI group. However, this was significantly elevated in the combined treatment group (P SP-C mRNA expression in lung tissues. The protein assay showed a decreased level of infiltration and activation of inflammatory cells, in addition to increased expression of SP-C, thereby confirming the efficacy of this treatment in preventing VILI during anesthesia.
Impact Factor an Index

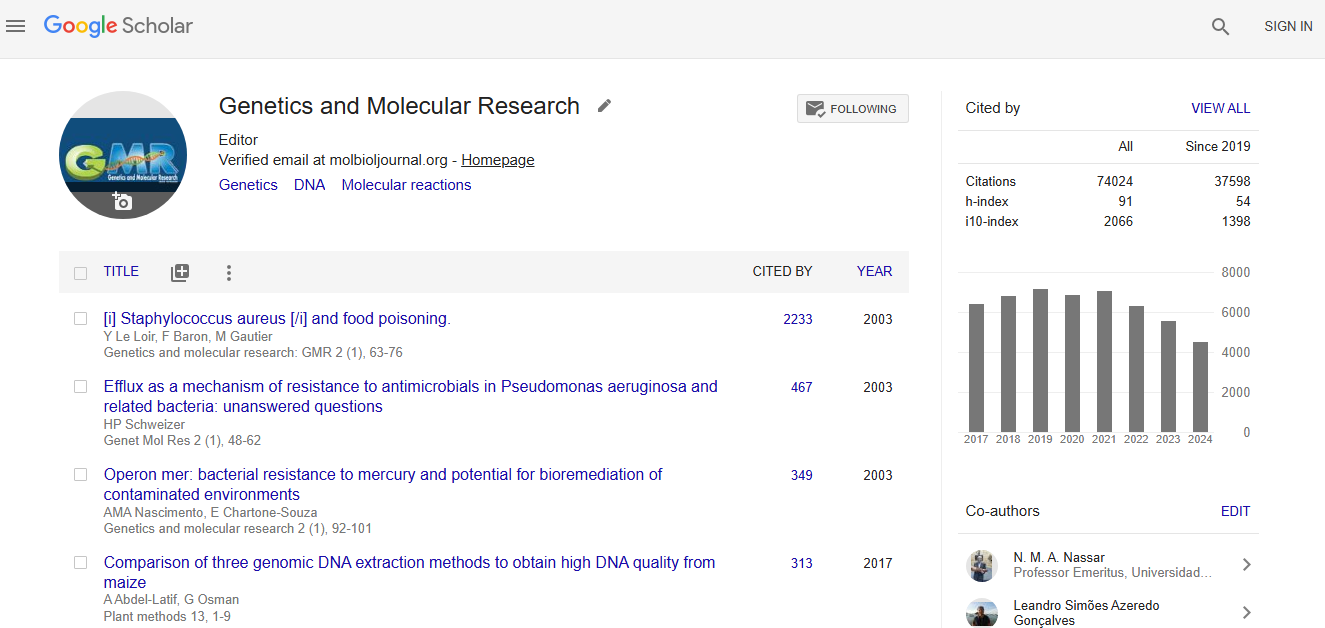
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report