Abstract
Identification of the isoamylase 3 gene in common wheat and its expression profile during the grain-filling period
Author(s): G.Z. Kang, G.Q. Liu, W. Xu, Y.J. Zhu, C.Y. Wang, H.Q. Ling and T.C. GuoIn higher plants, isoamylase-type starch debranching enzyme catalyzes the α-1,6-glucosidic linkages of glycogen and phytoglycogen. We cloned an isoamylase-type starch debranching enzyme ISA3 cDNA sequence (2883 bp), designated as TaISA3, from common wheat (Triticum aestivum), using the rapid amplification of cDNA ends method. The open reading frame of TaISA3 was found to have 2331 bp, and its deduced amino acid sequence was found to share high similarity with those of other gramineous plant ISA3 proteins. It contains a putative transit peptide (68 amino acids), N-terminus domain (107 amino acids), and a catalytic domain (173 amino acids). We extracted the expressed TaISA3 protein from Escherichia coli (BL21), and measured starch isoamylase activity. During the wheat grain-fi lling period, transcripts of the TaISA3 gene reached a maximum level at the early developmental stage, then declined, and increased again near the final maturation stage of the grain. We confirm that the ISA3 gene is present in common wheat; it appears to play a role in starch synthesis during early and late stages of the grain-filling period.
Impact Factor an Index

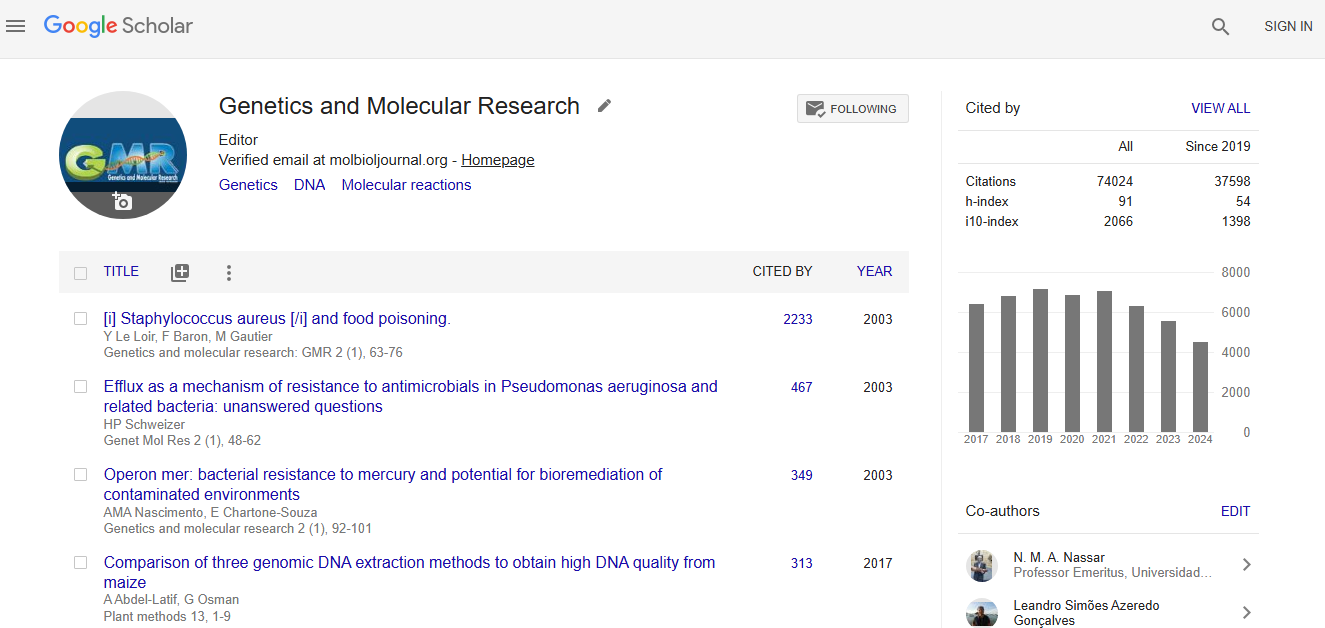
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report