Abstract
Genetic contribution of CYP2C9, CYP2C19, and APOE variants in acenocoumarol response
Author(s): J.A. Nastasi-Catanese1, J.R. Padilla-Guti├?┬®rrez2, Y. Valle2, F. Ortega-Guti├?┬®rrez3, M.P. Gallegos-Arreola4 and L.E. Figuera5,6Oral anticoagulants of the coumarin type have an inconveniently narrow therapeutic window, making their use difficult. In Mexico, genetic variables that participate in the heterogeneity of the therapeutic response remain poorly investigated. With the focus on warfarin, extensive pharmacogenomic studies have been performed, including those on the CYP450 family and APOE. The objective of this study was to determine the contribution of CYP2C9, CYP2C19, and APOE polymorphisms to the variations in response to the doses of acenocoumarol, which is the main anticoagulant prescribed to the Mexican population. The polymerase chain reaction–restriction fragment length polymorphism method was applied to identify *2 and *3 of CYP2C9, *2 of CYP2C19, and APOE variants. The genetic distribution of every polymorphism tested showed high variability when compared with other populations worldwide.
Impact Factor an Index

Google scholar citation report
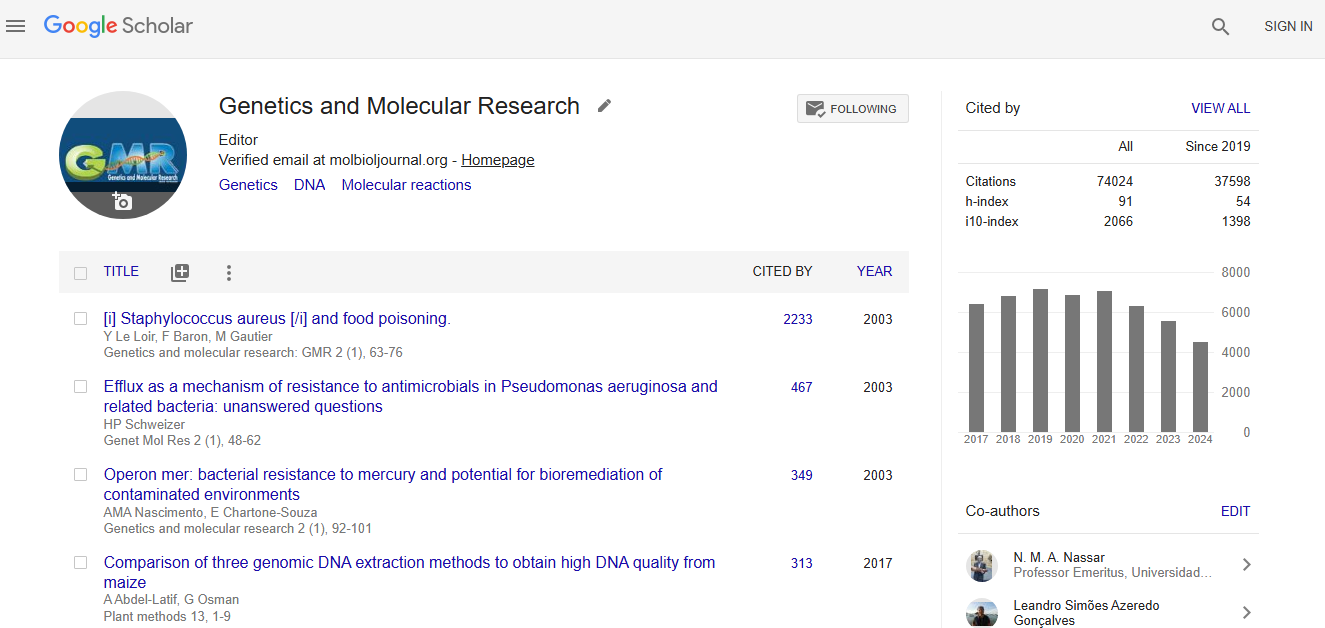
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report