Abstract
Full-length cDNA cloning and structural characterization of preproinsulin in Alligator sinensis
Author(s): R. Zhang, S.Z. Zhang, E. Li, C. Wang, C.L. Wang and X.B. WuInsulin is an important endocrine hormone that plays a critical physiological role in regulating metabolism and glucostasis in vertebrates. In this study, the complete cDNA of Alligator sinensis preproinsulin gene was cloned for the first time by reverse transcription-polymerase chain reaction and rapid amplification of cDNA ends methods; the amino acid sequence encoded and protein structure were analyzed. The full-length of preproinsulin cDNA sequence consists of 528 base pairs (bp), comprising a 34-bp 5'-untranslated region, a 170-bp 3'-untranslated region and an open reading frame that is 324 bp in length. The open reading frame encodes a 107-amino acid preproinsulin with a molecular weight of approximately 12,153.8 Da, theoretical isoelectric point of 5.68, aliphatic index of 92.06, and grand average of hydropathicity of -0.157, from which a signal peptide, a B-chain, a C-peptide, and an A-chain are derived. Online analysis suggested that the deduced preproinsulin amino acid sequence contains a transmembrane region, and that it has a signal peptide whose cleavage site occurs between alanine 24 and alanine 25. Comparative analysis of preproinsulin amino acid sequences indicated that the A-chain and B-chain sequences of preproinsulins are highly conserved between reptiles and birds, and that the preproinsulin amino acid sequence of Alligator sinensis shares 89% similarity to that of Chelonia mydas, but low similarity of 48-63% to those of mammals and fishes. The phylogenetic tree constructed using the neighbor-joining method revealed that preproinsulin of Alligator sinensis had high homology with reptiles and birds, such as Chelonia mydas, Gallus gallus, and Columba livia.
Impact Factor an Index

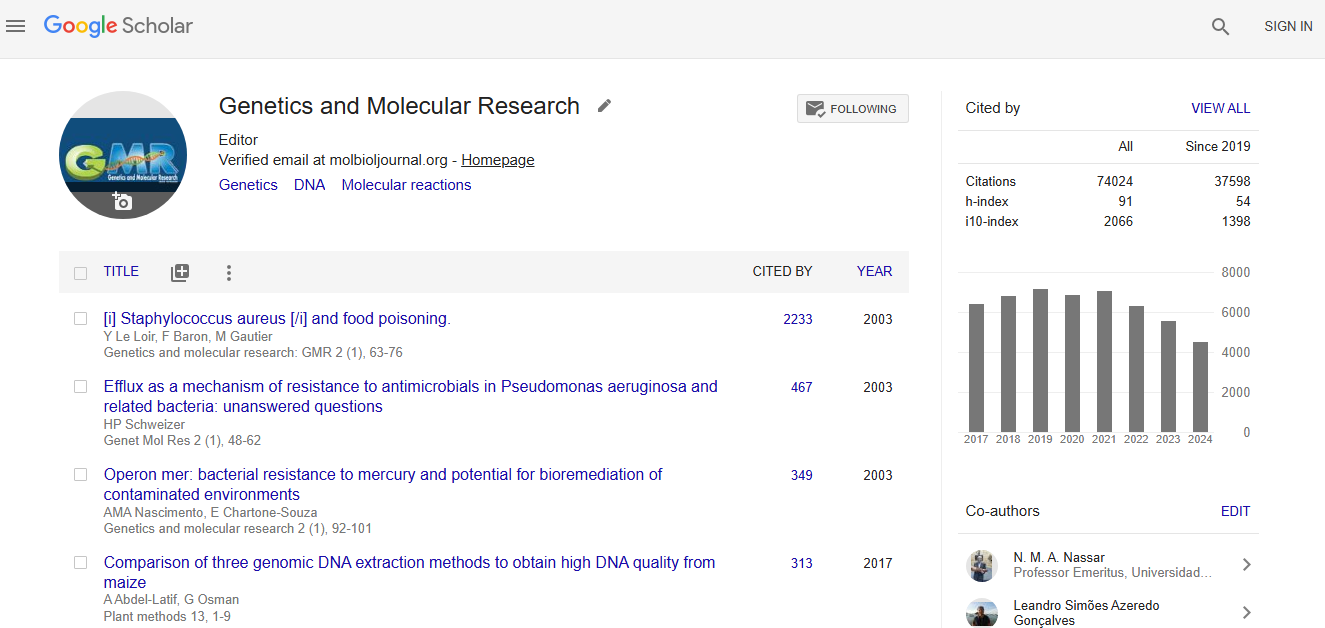
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report