Abstract
Expression of the bovine papillomavirus type 1, 2 and 4 L1 genes in the yeast Pichia pastoris
Author(s): A.L.S. Jesus, F.C. Mariz, H.M. Souza, M.N. Cordeiro, E.C. Coimbra, M.C.G. Leit├?┬Żo, L.M. Nascimento, R.C. Stocco, W. Be├?┬¦ak and A.C. FreitasPapillomaviruses are known to cause benign or malignant lesions in various animals. In cattle, bovine papillomavirus (BPV) is the etiologic agent of papillomatosis and neoplasia of the upper gastrointestinal tract and urinary bladder. Currently, there are no standard diagnostic tests or prophylactic vaccines. Protection against papillomavirus infection is conferred by neutralizing antibodies directed towards the major structural protein L1. These antibodies can be efficiently induced by immunization with virus-like particles that are formed spontaneously after L1 gene expression in recombinant systems. The yeast Pichia pastoris is known to provide an efficient system for expression of proteins due to reduced cost and high levels of protein production. We evaluated P. pastoris for expression of the L1 gene from BPV1, BPV2 and BPV4. After methanol induction, the recombinants were able to produce L1 proteins of the three different BPV types. To increase heterologous L1 protein levels, a codon optimization strategy was used for production under bioreactor conditions. The BPV1 L1protein was identified by monoclonal antibody anti-6xHis. This is the first report of BPV L1 expression in yeast.
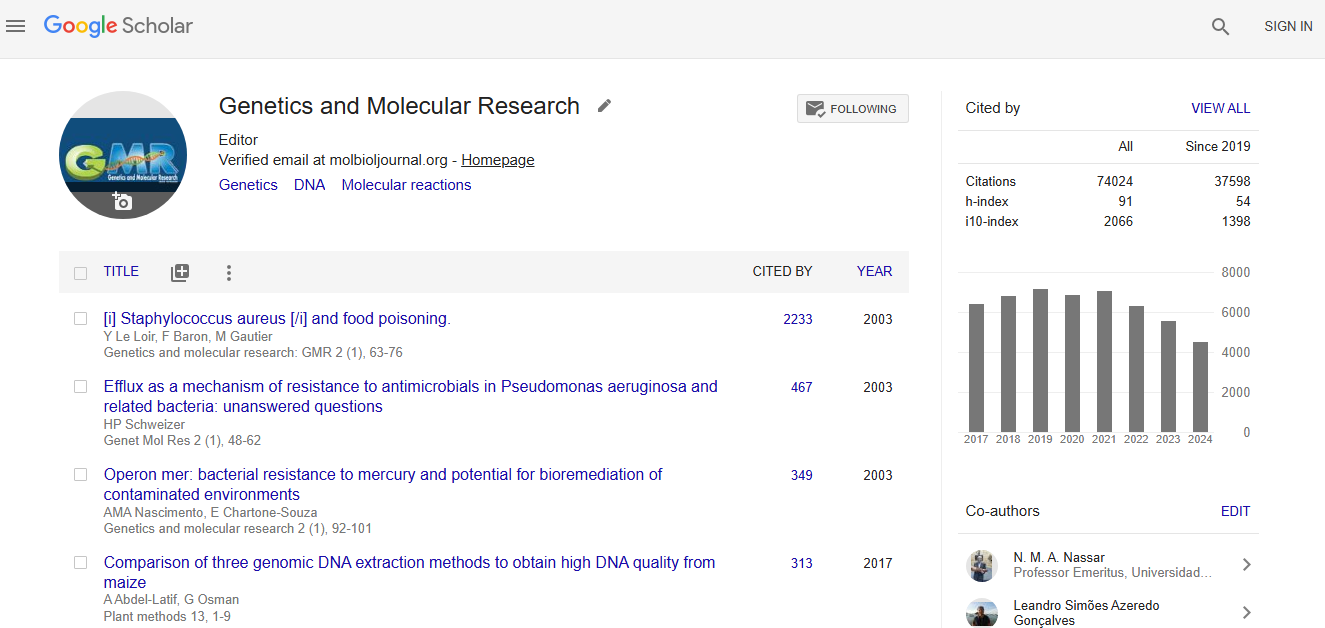
Impact Factor an Index

Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report