Abstract
Expression of heat shock protein 90 alpha (Hsp90�?±) in primary neonatal rat myocardial cells exposed to various periods of heat stress in vitro
Author(s): A. Islam, B. Rehana, M. Zhang, Z.J. Liu, S. Tang, J. Hartung and E.D. BaoThe objective of this study was to investigate the mechanism of heat shock protein 90 alpha (Hsp90α) protection against heart damage resulting from heat stress by detecting Hsp90α mRNA, Hsp90α protein, protein localization, and cell damage in primary myocardial cells of neonatal rats in response to heat stress in vitro. The cells were heat-stressed at 42°C in an incubator with 95% air and 5% CO2 for different periods. Levels of Hsp90α, protein localization, enzymes, and cytopathological lesions were detected using Western blot, immunocytochemistry enzymatic assays, and cytopathological techniques. Aspartate aminotransferase, lactate dehydrogenase, and creatine kinase enzyme levels were elevated during heat stress, and acute cellular lesions that were characterized by vacuolar degeneration and necrosis were observed. Hsp90α levels decreased between 10 and 60 min of heat stress and increased after 360 and 480 min, while Hsp90α mRNA decreased after 360 min. These results indicate that heat stress might induce irreversible damage in certain myocardial cells. The elevated Hsp90α level at the end of heat stress and its positive signal in the cytoplasm of myocardial cells after heat stress could be associated with its protective role. Additionally, the consumption of Hsp90α exceeded its production in the first period of treatment.
Impact Factor an Index

Google scholar citation report
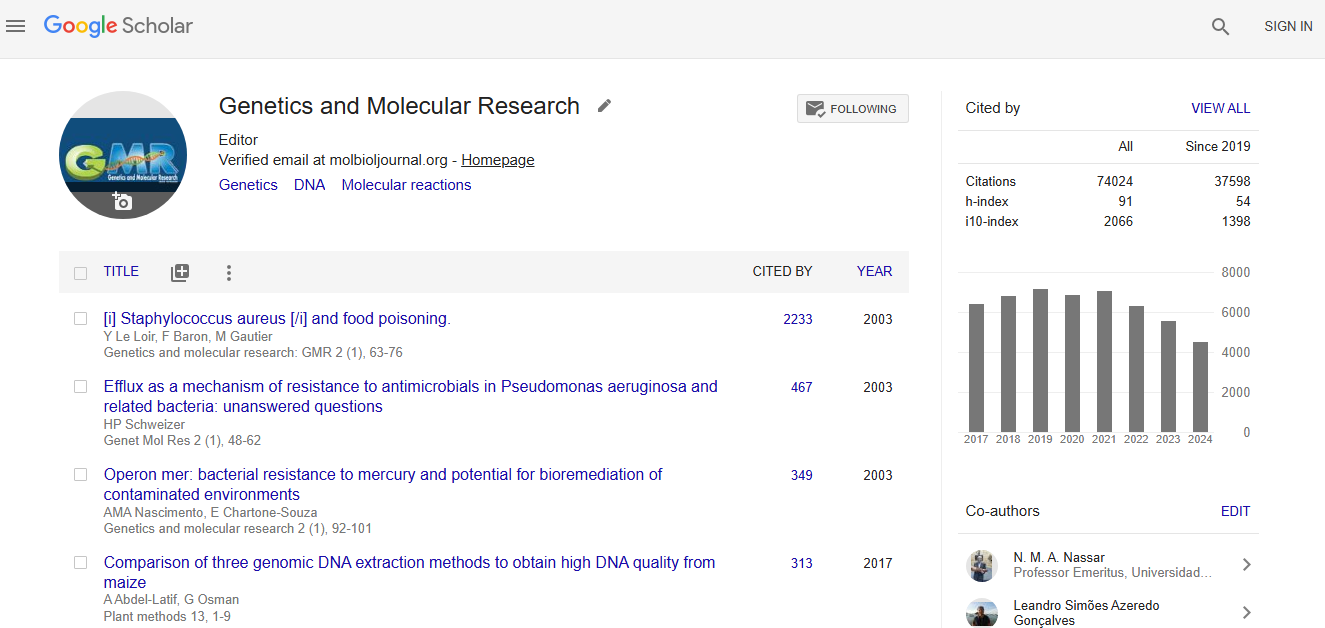
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report