Abstract
Effects of gemcitabine on radiosensitization, apoptosis, and Bcl-2 and Bax protein expression in human pancreatic cancer xenografts in nude mice
Author(s): Z.T. Shen, X.H. Wu, L. Wang, B. Li, X.X. ZhuThe aim of this study was to evaluate the radiosensitizing effects of gemcitabine towards human pancreatic cancer xenografts. A human pancreatic cancer xenograft model was established in nude mice, 36 of which were randomly divided into 6 treatment groups. Tumors were measured every 2 days, and the tumor volumes, growth delays, and inhibition rates were compared to evaluate the gemcitabine enhancement factor. The apoptotic index was determined by terminal deoxynucleotidyl transferase dUTP nick end-labeling assay, and apoptosis inhibitory protein Bcl-2 and apoptosis-related protein Bax expression were detected by immunohistochemistry. Compared with the control group, xenograft growth was significantly inhibited in the 25 (G25) and 50 mg/kg gemcitabine (G50) groups (P < 0.05). In the 25 (G25R) and 50 (G50R) mg/kg gemcitabine + radiotherapy groups, local tumor growth was significantly inhibited, with inhibition rates of 88.22 and 91.23%, respectively, significantly higher than those of the simple radiotherapy (SR), G25, and G50 groups (44.11, 72.88, and 77.53%, respectively; P < 0.05). The tumor growth delay in the G25R and G50R groups were 9 and 15 days, respectively, higher than the SR, G25, and G50 groups (each 4 days, P < 0.05). The apoptosis of tumor cells in the intervention groups significantly increased, and the apoptotic index among the intervention groups exhibited significant differences (P < 0.05). The immunohistochemical results indicated that Bcl-2 was downregulated to different degrees in the intervention groups, whereas Bax was upregulated (P < 0.05). Therefore, gemcitabine appears to enhance the radiotherapeutic sensitivity of human pancreatic cancer xenografts significantly.
Impact Factor an Index

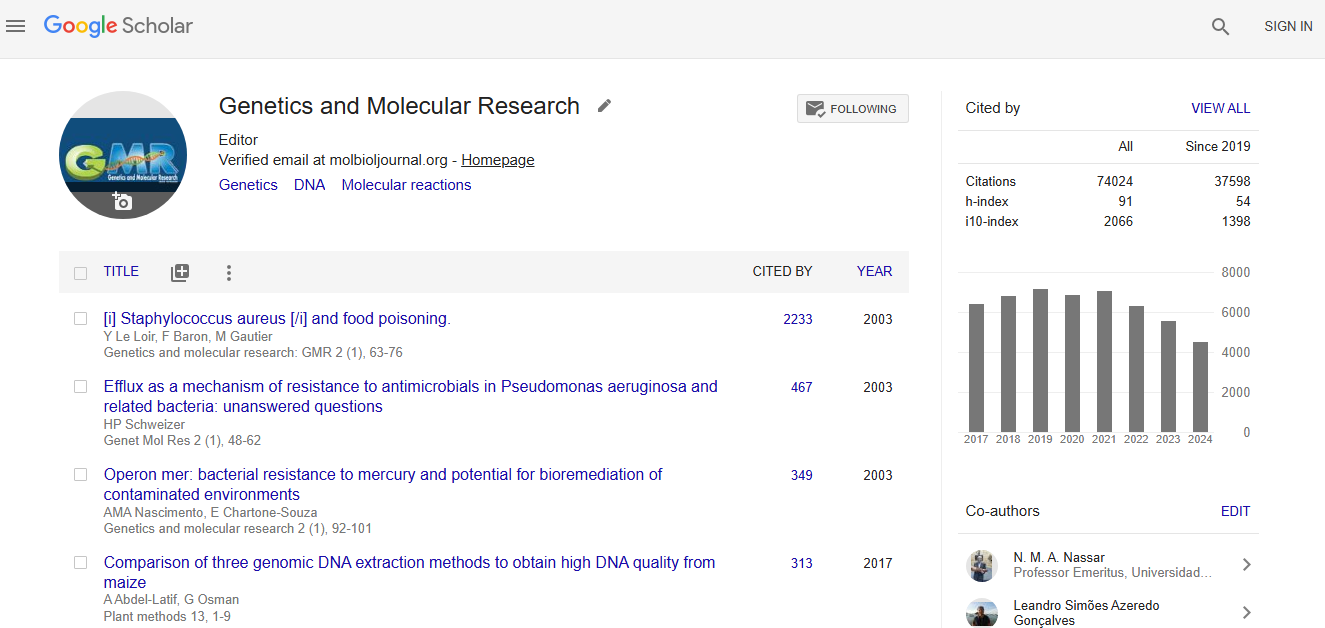
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report