Abstract
Effectiveness of olfactory ensheathing cell transplantation for treatment of spinal cord injury
Author(s): Y.J .Rao, W.X. Zhu, Z.Q. Du, C.X. Jia, T.X. Du, Q.A. Zhao, X.Y. Cao and Y.J. WangThe aim of this study was to determine the effectiveness and safety of transplantation of olfactory ensheathing cells for functional repair of the spinal cord. An olfactory bulb was obtained from a 4- to 5-month-old aborted fetus, and it was digested into single olfactory ensheathing cells and then cultured and purified for 1 to 2 weeks. Under general anesthesia, these single-cell suspensions of olfactory ensheathing cells were injected into the corresponding spinal injury site with 0.45-mm-diameter injections. The American Spinal Injury Association (ASIA) Impairment Scale was used to evaluate spinal function. A total of 15 patients (12 men, 3 women; age range, 18-56 years; mean age, 40) were admitted for obsolete spinal injuries. Spinal functions of the 15 patients were observed and followed postoperatively for a period ranging from 2 weeks to 1 month. All the 15 patients exhibited improvements in spinal function, and the improvement tendencies continued. Twelve patients had obvious spinal function improvement, and three had slight improvement according to the ASIA scale, with an obvious difference between preoperation and postoperation measures (P < 0.05). No fevers, infections, functional deteriorations, or deaths were seen. Thus, transplantation of olfactory ensheathing cells promoted spinal and neurofunctional recovery in patients with malignant spinal injuries, and this therapeutic method was safe.
Impact Factor an Index

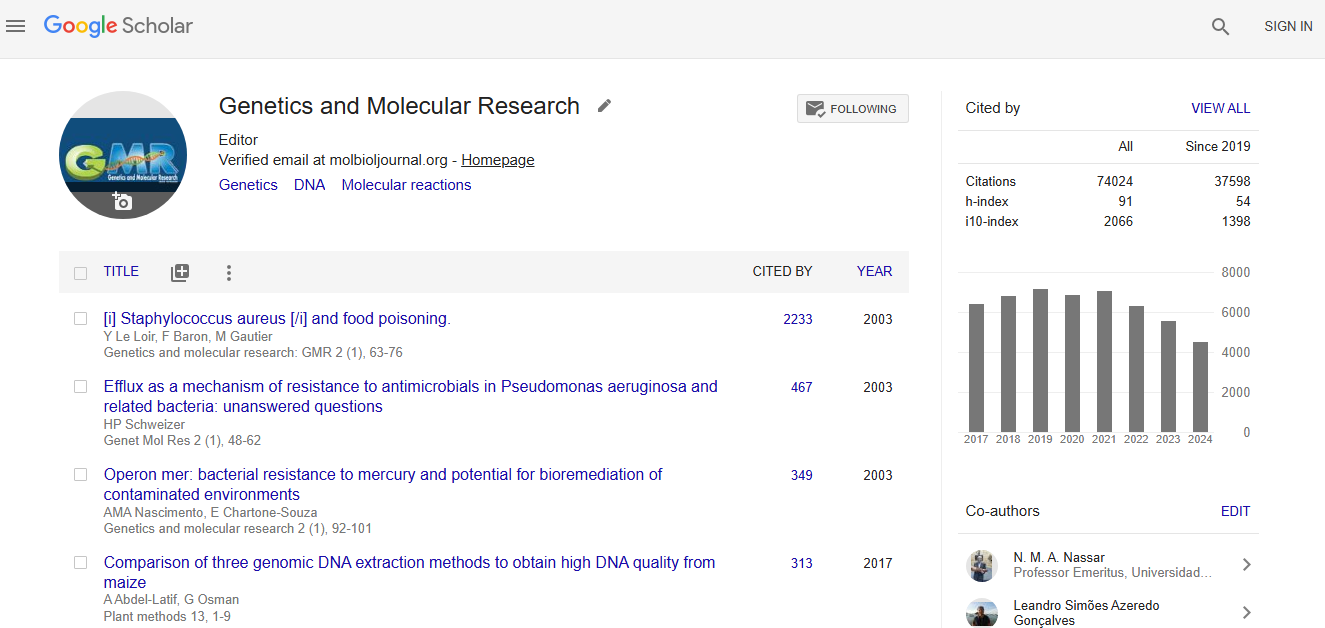
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report