Abstract
Effect of inhibition of MEK pathway on 5-aza-deoxycytidine-suppressed pancreatic cancer cell proliferation
Author(s): X. Wang1, H. Wang1, N. Jiang1, W. Lu1, X.F. Zhang1 and J.Y. Fang2Pancreatic adenocarcinoma is a lethal disease because it is inoperable at the time of diagnosis. Therefore, the search for new therapeutic approaches is critical. The abnormal expression of the mitogen-activated protein kinase/extracellular signal-regulated kinase (MEK) pathway and alteration in epigenetic modification (DNA methylation and acetylation of histones) is a common feature in the majority of human pancreatic adenocarcinomas. Because DNA methyltransferase levels are regulated by the MEK pathway, we examined the effects of an MEK inhibitor, PD98059, on the action of DNA methyltransferase inhibitor 5-aza-2'-deoxycytidine (5-aza-dC), the epigenetic agent in the pancreatic cell line CFPAC1. Our results showed that PD98059 significantly potentiated the capability of 5-aza-dC to induce a cessation of cell proliferation concomitant with cell cycle arrest. We also observed an increase in tumor suppressor gene expression associated with the efficacy of treatment with PD98059 and 5-aza-dC. Further studies explored the molecular mechanisms by which 5-aza-dC induced the expression of p21WAF1. We found that 5-aza-dC induced acetylation of histone H3 on the p21WAF1 gene promoter and demethylation status on the p21WAF1 gene promoter region. These effects were strikingly enhanced by the concomitant blockade of the MEK pathway. Furthermore, knockdown of p21WAF1 by small interfering RNA rescued human pancreatic cancer cells from 5-aza-dC-mediated growth inhibition. Taken together, our results show that the MEK inhibitor enhanced the effects of 5-aza-dC in human pancreatic cancer cells. These results suggest that the MEK signal pathway may be a potential target for pancreatic cancer therapy.
Impact Factor an Index

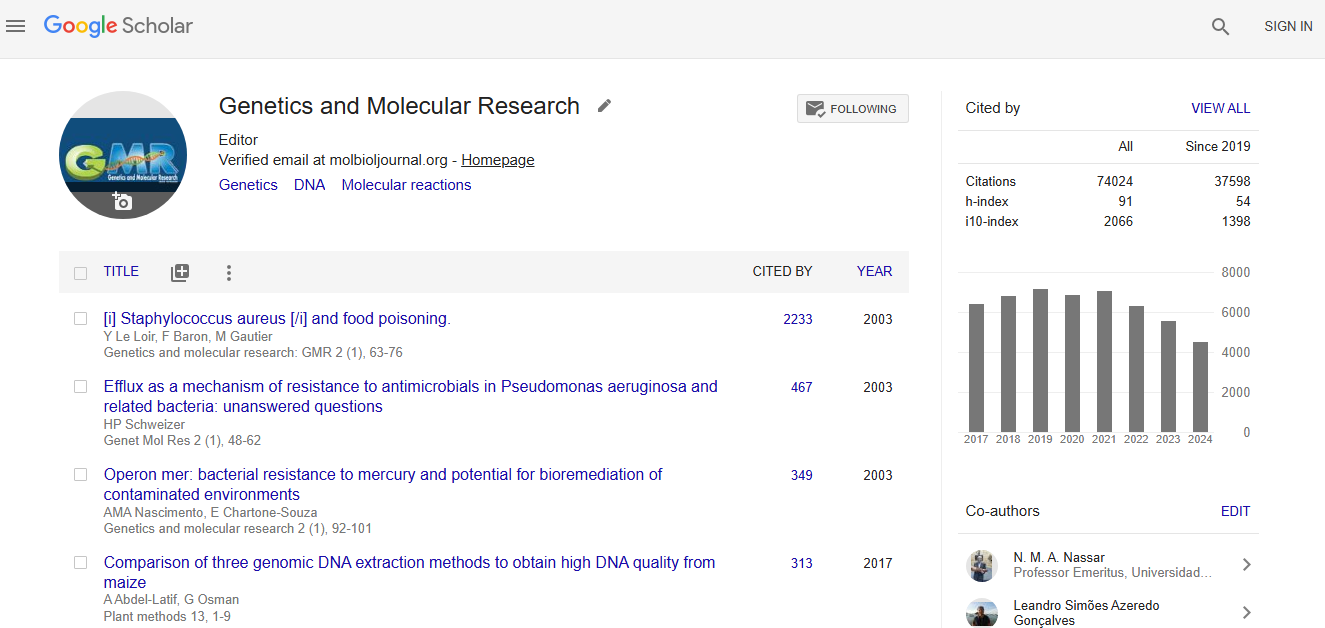
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report