Abstract
Comparison of physical and chemical characteristics of collagen from the skin of cod (Gadus macrocephaius)
Author(s): Y. Shu, H. Ren, R. Ao, W.C. Qi and Z.S. ZhangAcid and pepsin-soluble collagen (ASC and PSC, respectively) were extracted from cod (Gadus macrocephaius) skin, and yields of 37.36 and 55.96% were obtained for ASC and PSC, respectively. The total yield of ASC and PSC was 93.92%, based on the lyophilized dry weight, which is higher than that obtained from other sources. Electrophoresis revealed that both ASC and PSC consisted of two different α-chains (α1 and α2), which were characterized as type I collagen. Analysis of amino acids showed that both the ASC and PSC contained imino acids (216.1 and 190.6 residues/1000 residues, respectively) and the Fourier transform infrared spectroscopy spectra of both collagens were similar with pepsin hydrolysis having no effect on their triple-helical structure. The thermal denaturation temperature of ASC and PSC, as measured by viscometry, was 26.8° and 25.6°C, respectively.
Impact Factor an Index

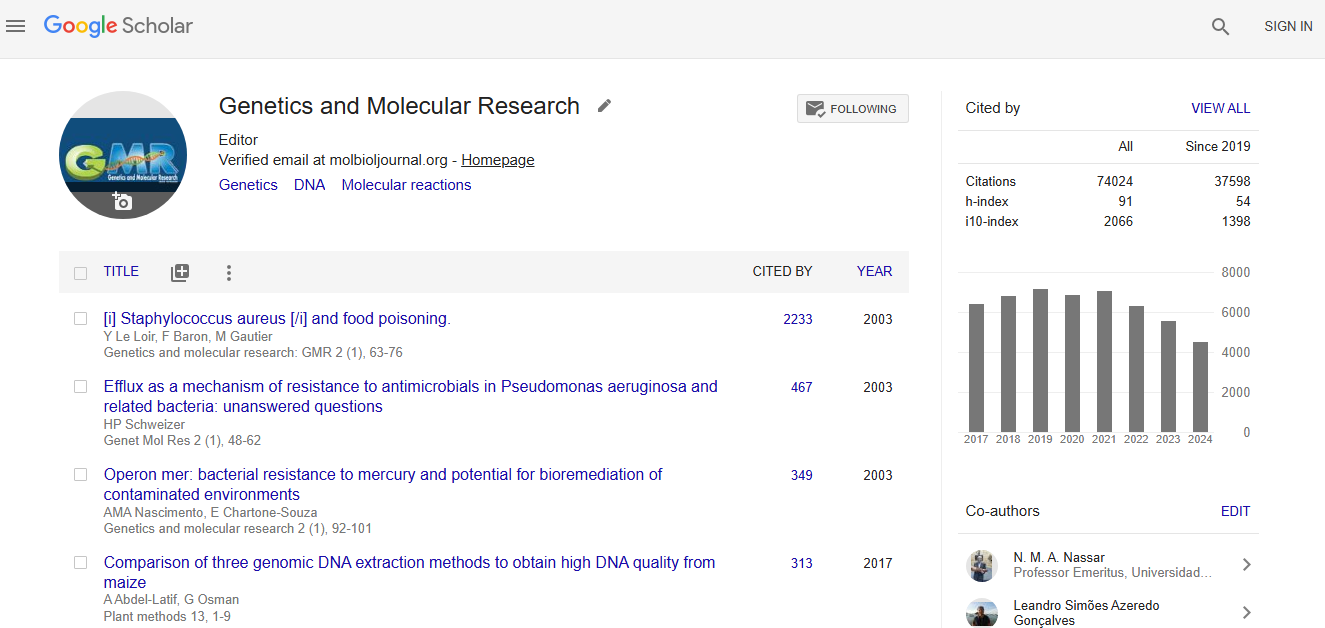
Google scholar citation report
Citations : 74024
Genetics and Molecular Research received 74024 citations as per google scholar report